Scientists develop parasitic worms to hunt and destroy cancer cells
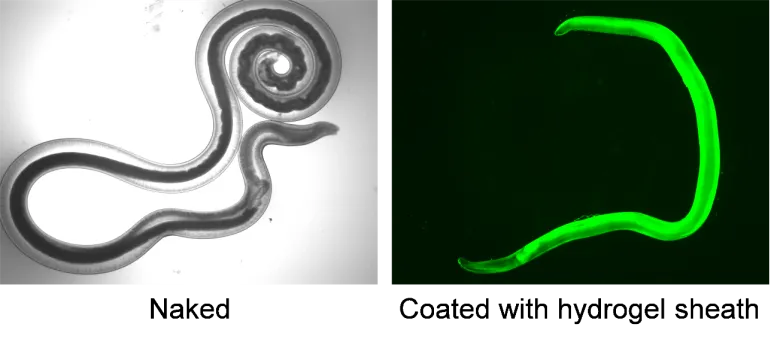
- Osaka University researchers have developed hydrogel-coated nematodes capable of delivering anti-cancer drugs directly to tumors.
- The parasitic worm Anisakis simplex naturally senses cancer odors, enabling precise targeting in oncology breakthroughs.
- In vitro experiments demonstrate the worms’ ability to protect themselves while eliminating malignant cells in targeted therapy.
In the relentless battle against cancer, where traditional chemotherapy often ravages healthy tissues alongside tumors, a groundbreaking innovation from Japan is turning heads in the oncology community.
Scientists at Osaka University have harnessed the unlikely power of parasitic worms—creatures typically associated with foodborne illnesses—to create a potential new weapon in precision medicine.
These engineered nematodes, cloaked in custom hydrogel armor, could revolutionize how we approach tumor treatment by delivering lethal payloads straight to the heart of malignant growths.
The story begins with Anisakis simplex, a marine nematode infamous for causing anisakiasis in humans who consume raw or undercooked seafood.
But this worm has a hidden talent: it can detect the subtle chemical signatures, or “odors,” emitted by cancer cells.
Previous studies have shown that certain nematodes, including related species like Caenorhabditis elegans, exhibit chemotaxis—a directed movement toward specific scents.
In cancer detection research dating back to 2015, Japanese scientists discovered that C. elegans could identify pancreatic cancer through urine samples with remarkable accuracy, paving the way for non-invasive diagnostics.
Building on this, the Osaka team wondered: could these worms do more than just sniff out danger? Could they be weaponized against it?
Led by biomedical engineer Shinji Sakai and first author Wildan Mubarok, the researchers set out to transform these microscopic predators into drug-delivery vehicles.
Their approach involves a sophisticated bioengineering technique where the worms are dipped into a sequence of solutions containing tyramine-modified carboxymethyl cellulose and hydrogen peroxide.
This triggers an enzymatic reaction mediated by horseradish peroxidase anchored to the nematode’s cuticle, forming a flexible hydrogel sheath around the worm in just 20 minutes.
The resulting coating, a mere 0.01 millimeters thick, acts like a tailored suit—protective, customizable, and capable of carrying therapeutic cargos.
What makes this hydrogel technology stand out in the field of nanomedicine?
Unlike rigid nanoparticles or liposomes that struggle with biological barriers, the sheath is biocompatible and elastic, ensuring the worm retains full motility.
Tests revealed that encased nematodes swam just as vigorously as their naked counterparts, navigating toward attractive chemical gradients without hindrance.
Moreover, the coating shielded them from harsh environmental threats, such as ultraviolet radiation and oxidative stress from hydrogen peroxide, which could otherwise kill the worms before they reach their target.
But the real excitement lies in the payload. The team loaded the sheaths with anti-cancer agents, including doxorubicin, a potent chemotherapy drug known for its efficacy against various tumors but also its severe side effects like cardiotoxicity when administered systemically.
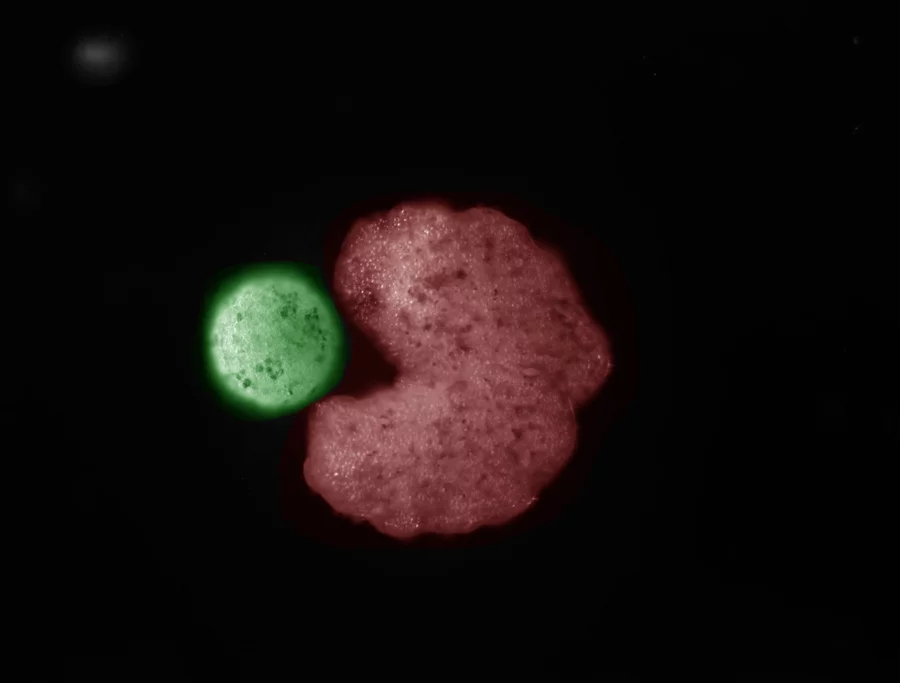
In laboratory dishes, these armored worms zeroed in on cultured cancer cells, releasing their deadly cargo upon contact.
Microscopy footage captured the dramatic moment: nematodes attaching to tumor cells, the hydrogel degrading in the acidic microenvironment typical of malignancies, and the drugs infiltrating to trigger apoptosis—programmed cell death.
This isn’t the first time parasites have been eyed for medical use. Historical anecdotes trace back to ancient practices where leeches and maggots aided wound healing, but modern biotechnology elevates this to a new level.
In oncology, targeted therapies like antibody-drug conjugates have shown promise, yet they often fail to penetrate solid tumors effectively.
The nematode system addresses this by leveraging the worm’s innate tropism—the biological urge to seek out cancer-specific cues, possibly volatile organic compounds released by hypoxic tumor tissues.
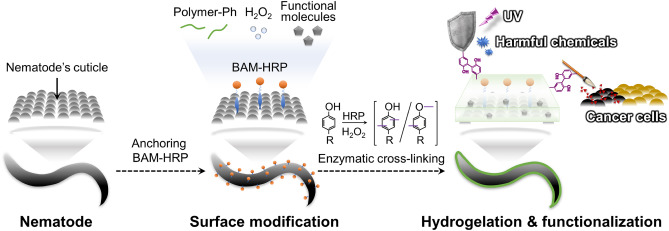
As the experiments progressed, the researchers uncovered intriguing nuances.
For instance, the hydrogel’s composition could be fine-tuned: incorporating fluorescent dyes for imaging or even antimicrobial peptides for dual-action against infections in immunocompromised patients.
In one trial, sheathed worms survived exposure to conditions that obliterated uncoated ones, hinting at applications beyond cancer—perhaps delivering probiotics to gut microbiomes disrupted by aggressive treatments.
Yet, how exactly do these worms “smell” cancer?
The mechanism involves olfactory receptors on the nematode’s sensory organs, tuned to metabolites like 2-ethyl-1-hexanol, a compound overexpressed in certain carcinomas.
This sensory prowess, evolved over millions of years for host-finding in marine environments, now serves a therapeutic purpose.
The Osaka team’s in vitro success raises tantalizing questions: could this translate to living organisms?
| Aspect | Details |
|---|---|
| Nematode Species | Anisakis simplex |
| Hydrogel Sheath Thickness | 0.01 mm |
| Coating Time | 20 minutes |
| Protective Functions | Shields from UV light and hydrogen peroxide |
| Drug Delivery Method | Loaded with anti-cancer agents like doxorubicin |
| Targeting Mechanism | Chemotaxis toward cancer odors |
| Publication | Materials Today Bio, June 2022 |
Diving deeper into the methodology, the process starts with immobilizing the worms in a cold saline solution to minimize stress during coating.
The enzymatic cross-linking ensures the sheath adheres seamlessly to the cuticle without penetrating internal tissues, preserving the nematode’s viability for up to several days.
Viability assays showed over 90% survival rates post-encapsulation, a critical factor for any living delivery system.
The implications for clinical oncology are profound.
Imagine injecting these bioengineered worms into a patient’s bloodstream, where they migrate to metastatic sites undetected by the immune system, thanks to the hydrogel’s stealth properties.
This could slash the required drug dosage, minimizing neuropathy and nausea that plague conventional regimens.
In prostate cancer or melanoma, where tumors are accessible, localized delivery might enhance immunotherapy synergies, boosting T-cell infiltration.
But challenges loom. Anisakis simplex’s parasitic nature raises safety concerns—could it trigger allergic reactions in sensitive individuals?
The team mitigated this by selecting non-reproductive stages and ensuring the sheath prevents host invasion.
Animal trials, though not yet detailed in public releases, are the next frontier, with rodent models testing efficacy against xenograft tumors.
Expanding the scope, this technology intersects with emerging fields like biohybrid robotics, where living organisms merge with synthetic materials.
Could we program these worms further, perhaps with genetic edits to enhance specificity for breast cancer or leukemia?
International collaborations are already buzzing, with experts in hydrogel biomaterials exploring scalable production.
As researchers refine the sheath’s degradation kinetics—ensuring drugs release only at tumor pH levels—the potential grows.
What if these worms could carry CRISPR tools for gene editing within tumors, correcting mutations at the source?
Or monitor treatment response in real-time via embedded biosensors?
The journey from lab bench to bedside is fraught with regulatory hurdles, but early indicators are promising.
With cancer claiming millions of lives annually, this worm-based strategy might just wriggle its way into the arsenal of next-generation therapies.
And as trials advance, one can’t help but wonder: will these tiny assassins redefine survival rates in the deadliest forms of the disease?